
Leave message
Can’t find what you’re looking for?
Fill out this form to inquire about our custom protein services!
Inquire about our Custom Services >>


































 Request a FREE Sample of our FcRn Binding Kit!
Request a FREE Sample of our FcRn Binding Kit! Request a FREE Sample of our Fc gamma RI / CD64 Binding Kit !
Request a FREE Sample of our Fc gamma RI / CD64 Binding Kit !
 Happy Holiday! Limited Keychain here with your next order
Happy Holiday! Limited Keychain here with your next order Happy Holiday! Limited Keychain here with your next order
Happy Holiday! Limited Keychain here with your next order
 Request a FREE sample of our GMP products!
Request a FREE sample of our GMP products!  Request a FREE sample of our GMP products!
Request a FREE sample of our GMP products!
> Bispecific antibody targets and services 
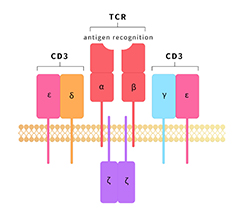
Bispecific antibodies (bsAbs) harness the specificities of two antibodies and combine them to simultaneously recognize different antigens or epitopes. This 'two-target' functionality has meant that interest in their use for therapeutic applications has increased considerably. With the development and maturity of bsAb technology platform, the combination selection of targets is becoming the key point of competition. Among the bsAb programs under development currently, the combination of CD3 and tumor surface targets are the most popular targets pairs. And the new targets of Immune cell will also provide more choice in targets combination in the future.
Free samples for bsAb hot targets are available!
> MALS verified heterodimer
> High bioactivity and batch-to-batch consistency
> Bioactivity verified by clinical bispecific antibody
> Various tags and species

> High purity and homogeneity
> High bioactivity verified
> Various tags and species
> High purity and homogeneity
> High bioactivity verified
> Suitable for immunization and inhibitor
> Authentic conformation
> Super bioactivity verified
> Exclusive Nanodisc technology
The characterization and pharmacokinetic (PK)/PD assessment are vital. Quality attributes such as antigen specificity; affinity and on- and off-rates; avidity (for bispecific antibodies that target two molecules on the same cell); potency; process-related impurities such as aggregates, fragments, and homodimers; stability; and half-life may affect pharmacology and should be studied. On the other hand, as bispecific antibodies may present as a mixture of biologically active and inactive forms, it is important to identify the bispecific antibody form(s) that is most pharmacologically relevant to PK/PD assessment and to develop validated assays that measure the appropriate form(s) accordingly. Due to the synergetic effect BsAb brings, the dosage is relatively low. Therefore, it requires a more sensitive assay for analysis.

Fig 1. Immobilized Human CD3E&CD3D Heterodimer Protein, His Tag&Tag Free (Cat. No. CDD-H52W1) at 2 μg/mL, add increasing concentrations of Bispecific T cell Engager (CD3 X BCMA) and then add Biotinylated BCMA Fc,Avitag (Cat. No. BC7-H82F0) at 0.2 μg/mL. Detection was performed using HRP-conjugated streptavidin with sensitivity of 4 ng/mL.


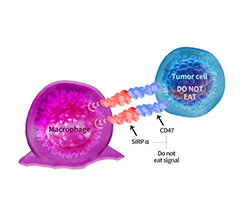
Fig 2. Binding assay between bsAb and Fc receptor

Fig 3. Immobilized Human OX40 Protein, His Tag (MALS verified) (Cat. No. OX0-H5224) at 2 μg/mL, add increasing concentrations of CTLA-4 x OX40 bispecific antibody in 50% Human serum and then add Biotinylated Human CTLA-4, Fc,Avitag (Cat. No. CT4-H82F3) at 0.2 μg/mL. Detection was performed using HRP-conjugated streptavidin with sensitivity of 4 ng/mL (Intact assay)

Learn more about Bioactivity analysis of bispecific antibody
With a strong team of scientists, ACROBiosystems also provides high-quality integrated SPR&BLI analytical services. ACRO has served and collaborated with more than 100 customers from academia and industry. Our work supported multiple antibody drugs IND applications.
Click to learn more about SPR /BLI analytical service
This web search service is supported by Google Inc.









