 Request a FREE Sample of our FcRn Binding Kit!
Request a FREE Sample of our FcRn Binding Kit! Request a FREE Sample of our Fc gamma RI / CD64 Binding Kit !
Request a FREE Sample of our Fc gamma RI / CD64 Binding Kit !
 Happy Holiday! Limited Keychain here with your next order
Happy Holiday! Limited Keychain here with your next order Happy Holiday! Limited Keychain here with your next order
Happy Holiday! Limited Keychain here with your next order
 Request a FREE sample of our GMP products!
Request a FREE sample of our GMP products!  Request a FREE sample of our GMP products!
Request a FREE sample of our GMP products!
Designed under ISO 9001:2015 and ISO 13485:2016
Manufactured and QC tested under a GMP compliance factory
Animal-Free materials
Beta-lactam materials free
Batch-to-batch consistency
Stringent quality control tests
No animal derived peptone and lactose used in production process

This protein carries no "tag".
The protein has a calculated MW of 18.2 kDa. The protein migrates as 17 kDa±3 kDa when calibrated against Star Ribbon Pre-stained Protein Marker under reducing (R) condition (SDS-PAGE).
>95% as determined by SDS-PAGE.
Lyophilized from 0.22 μm filtered solution in PBS, pH7.4 with protectants.
Contact us for customized product form or formulation.
This product is supplied and shipped with blue ice, please inquire the shipping cost.
Upon receipt, store it immediately at -20°C or lower for long term storage.
Please avoid repeated freeze-thaw cycles.
This product is stable after storage at:
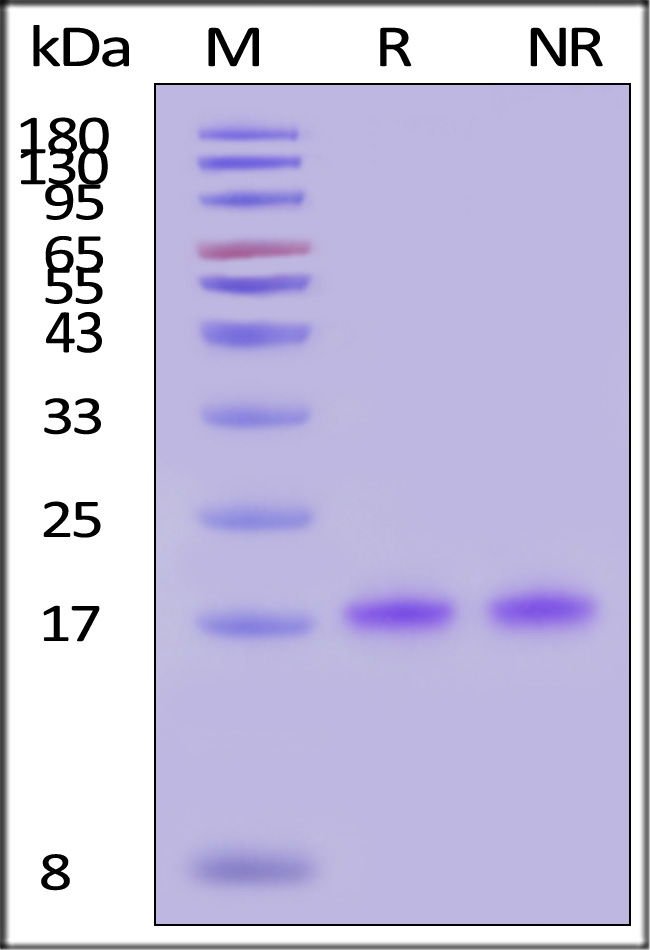
GMP Human IL-18 Protein on SDS-PAGE under reducing (R) and non-reducing (NR) conditions. The gel was stained with Coomassie Blue. The purity of the protein is greater than 95% (With Star Ribbon Pre-stained Protein Marker).
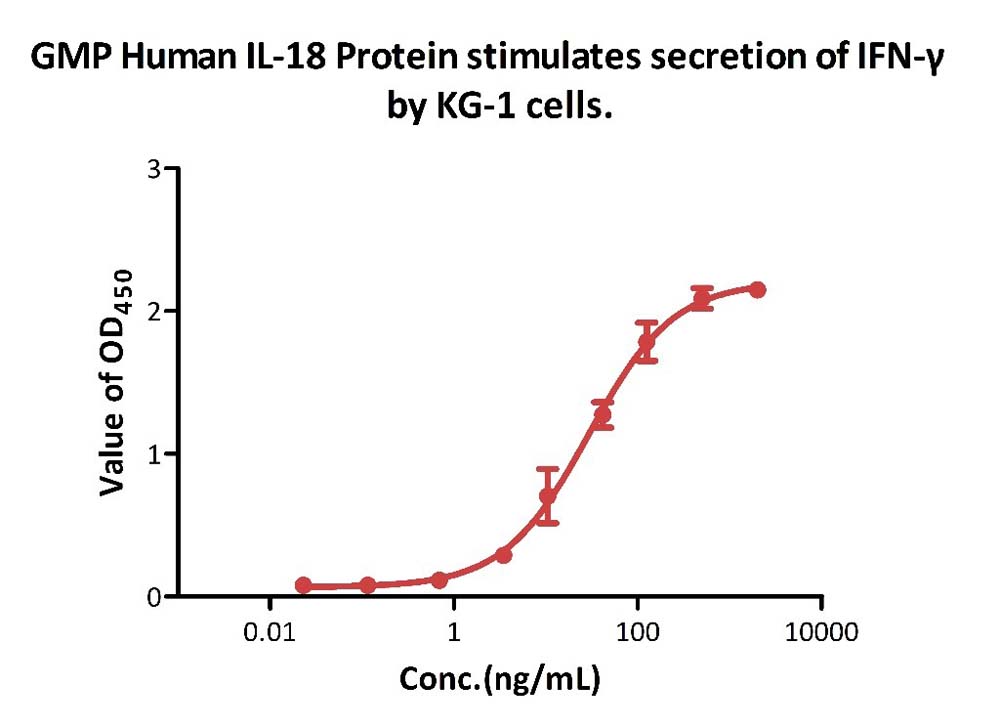
GMP Human IL-18 Protein (Cat. No. GMP-L18H16) stimulates secretion of IFN-γ by KG-1 cells. The specific activity of GMP Human IL-18 Protein is > 3.00ⅹ10^6 IU/mg, which is calibrated against WHO Reference Reagent Interleukin-18 (Human rDNA derived) (NIBSC code: 03/200) (QC tested).
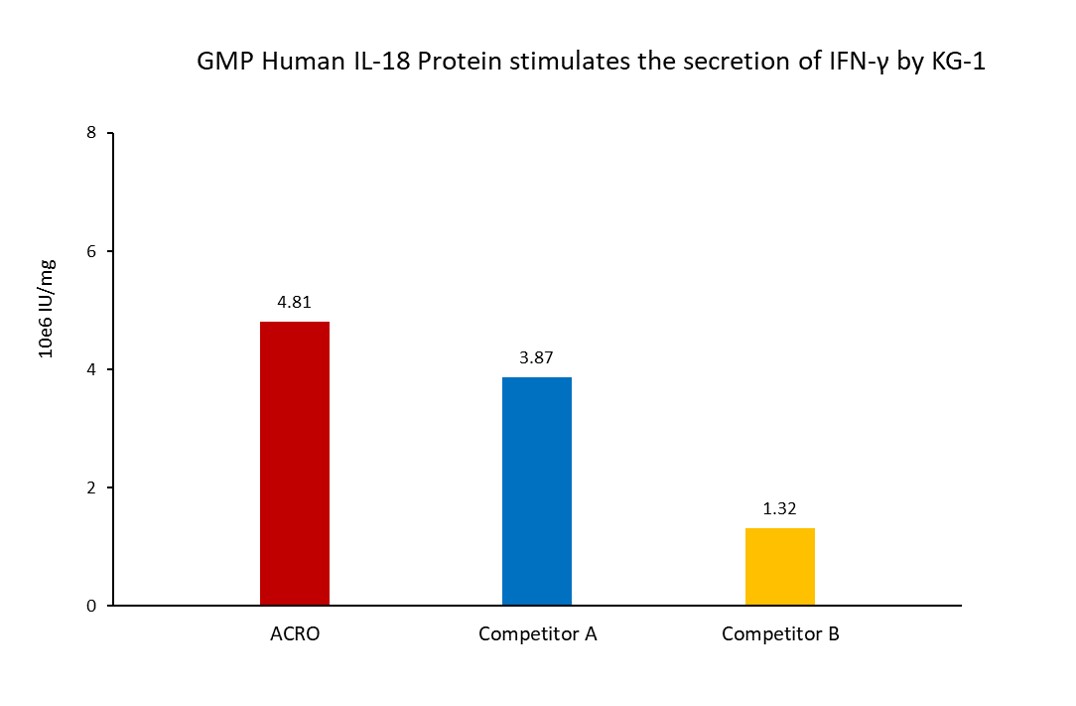
The activity of GMP Human IL-18 Protein (Cat. No. GMP-L18H16) was higher than other competing products.
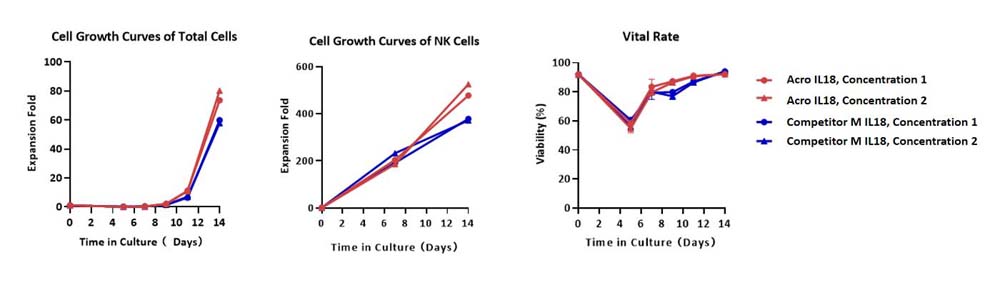
Human PBMCs were cultured with GMP Human IL-2 Protein (ACROBiosystems, Cat. No. GMP-L02H14), GMP Human IL-18 Protein (ACROBiosystems, Cat. No. GMP-L18H16) and GMP Human 4-1BB Ligand Protein (ACROBiosystems, Cat. No. GMP-41LH26), in CelThreaTM GMP T Cell Expansion Medium (ACROBiosystems, Cat. No. GMP-CM3101 & GMP-CM3101-1) for two weeks. The result shows that GMP Human IL-18 Protein can promote the expansion of these cells with a reasonable cell viability. Notably, the cells exhibit better expansion in GMP Human IL-18 Protein (ACROBiosystems).
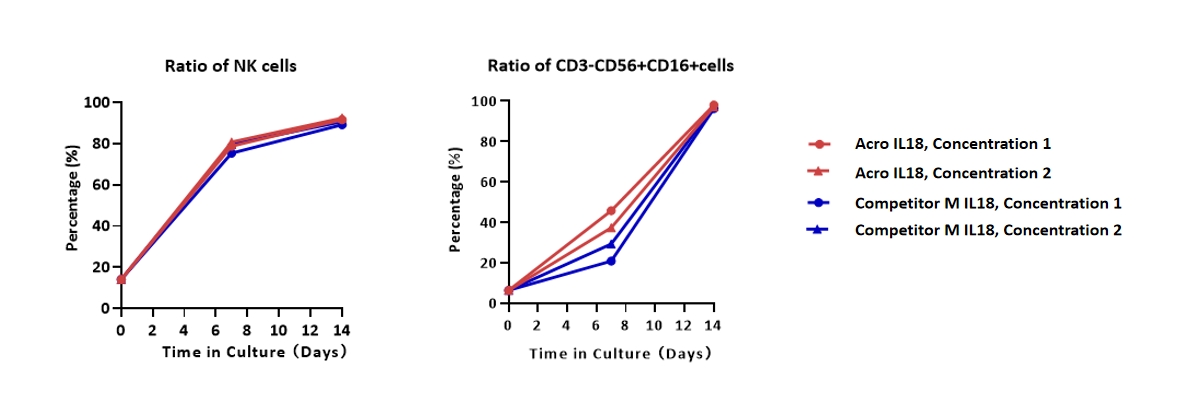
Human PBMCs were cultured with GMP Human IL-2 Protein (ACROBiosystems, Cat. No. GMP-L02H14), GMP Human IL-18 Protein (ACROBiosystems, Cat. No. GMP-L18H16) and GMP Human 4-1BB Ligand Protein (ACROBiosystems, Cat. No. GMP-41LH26), in CelThreaTM GMP T Cell Expansion Medium (ACROBiosystems, Cat. No. GMP-CM3101 & GMP-CM3101-1) for two weeks. The result shows that GMP Human IL-18 Protein (ACROBiosystems) can be comparable with the Competitor M IL-18 protein.
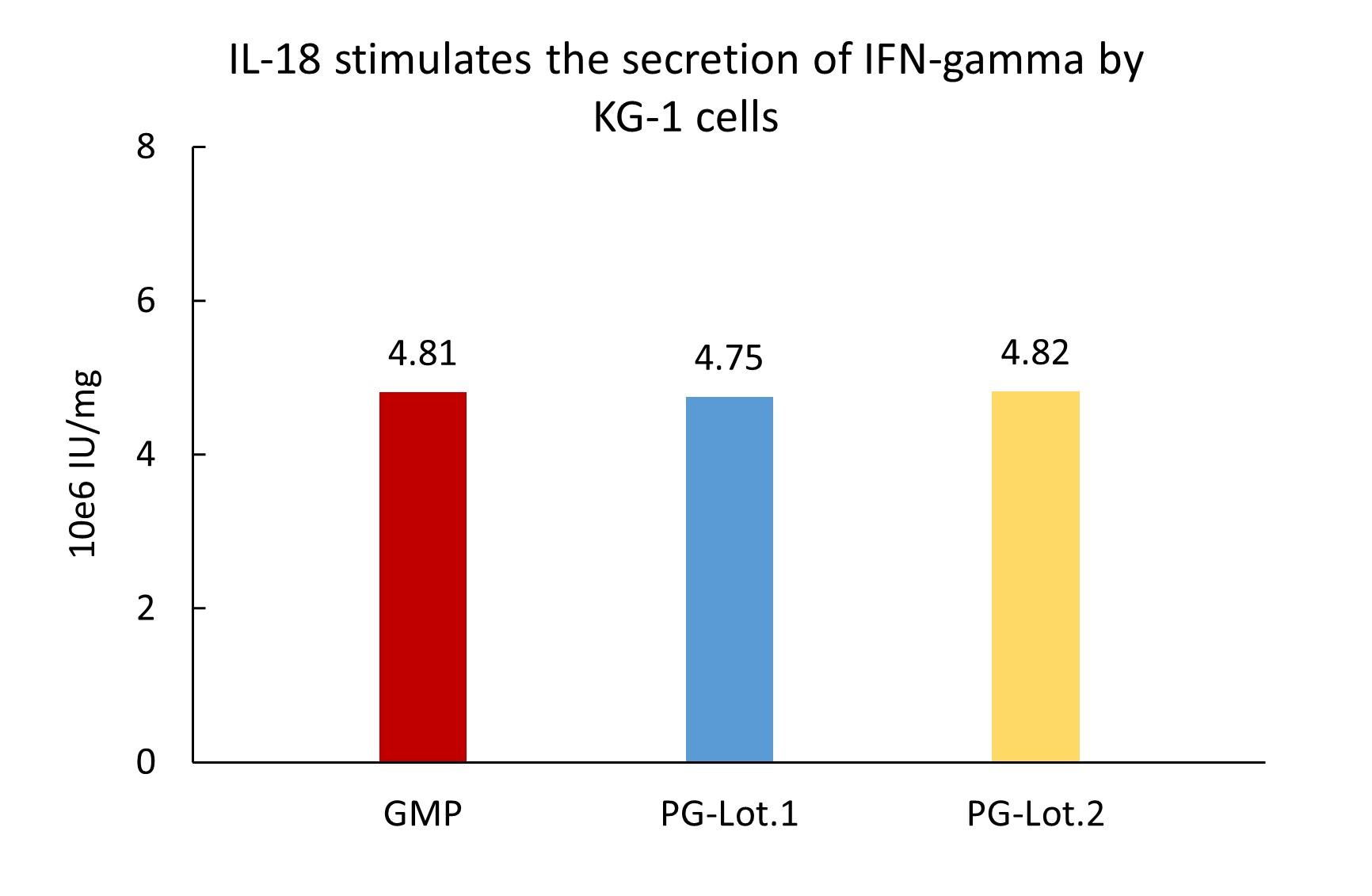
The Cell based assay shows batch-to-batch consistency between Acro's GMP and PG IL-18.
ACROBiosystems GMP grade products are produced under a quality management system and in compliance with relevant guidelines: Ph. Eur General Chapter 5.2.12 Raw materials of biological origin for the production of cell-based and gene therapy medicinal products; USP<92>Growth Factors and Cytokines Used in Cell Therapy Manufacturing; USP<1043>Ancillary Materials for Cell, Gene, and Tissue-Engineered Products; ISO/TS 20399-1:2018, Biotechnology - Ancillary Materials Present During the Production of Cellular Therapeutic Products.
ACROBiosystems Quality Management System Contents:
Designed under ISO 9001:2015 and ISO 13485:2016, Manufactured and QC tested under a GMP compliance factory.
Animal-Free materials
Materials purchased from the approved suppliers by QA
ISO 5 clean rooms and automatic filling equipment
Qualified personnel
Quality-related documents review and approve by QA
Fully batch production and control records
Equipment maintenance and calibration
Validation of analytical procedures
Stability studies conducted
Comprehensive regulatory support files
Request For Regulatory Support Files(RSF)
ACROBiosystems provide rigorous quality control tests (fully validated equipment, processes and test methods) on our GMP grade products to ensure that they meet stringent standards in terms of purity, safety, activity and inter-batch stability, and each bulk QC lot mainly contains the following specific information:
SDS-PAGE
Protein content
Endotoxin level
Residual Host Cell DNA content
Residual Host Cell Protein content
Biological activity analysis
Microbial testing
Mycoplasma testing
In vitro virus assay
Batch-to-batch consistency
Price(USD) : $1148.00
Price(USD) : $5875.00
Price(USD) : $9180.00

Explore our catalog of therapeutic antibody solutions to find the right products for you! We are dedicated to delivering solutions designed to help you drive innovation and push the boundaries of what therapeutic antibodies can be.

We offer a wide range of cell and gene therapy solutions starting from discovery to the clinic. Explore our wide range of proteins, antibodies, kits, and other assays to accelerate the development of your cell and gene therapy.

Organoid Toolbox is a collection of organoid solutions including ready-to-use organoids, organoid differentiation kits, and a variety of services to accelerate the progress of your drug development project.

ACROBiosystems developed a series of GMP grade cytokines under the GMP grade quality management system. Those products are all suitable for T/NK cell generation, activation, and proliferation in cell therapy research.

50+ targets designed for CAR detection, including PE/FITC/biotin labeled proteins. The key reagents for CD19 and BCMA were FDA DMF filed which can support your IND, NDA and BLA process.

Full length multi-pass TPs with stabilized structure and high bioactivity for immunization, antibody screening, cell based assay and CAR detection, including hot CD20, Claudin 18.2, CD133, GPRC5D,CCR8, CCR5, etc.

GMP grade cytokines, reagents for cell activation, gene edition, DNA/RNA removal, etc. Particularly focus on product design, quality control and solution-based support to link each phase of your cell and gene therapy journey.

A series of immune checkpoints including classic co-inhibitory and co-stimulatory receptors. The comprehensive catalog contains 100+ targets with various species and tags, and the high-quality proteins are in good batch-to-batch consistency.

To meet the needs of ADCs development, ACROBiosystems can provide: A variety of high-quality target proteins; MMPs/Cathepsin/uPA for cleavable linker; Anti-payload antibodies & anti-idiotypic antibodies for immunogenicity and PK analysis; SPR/BLI analytical and ADA development service.

Comprehensive collection of Fc receptor proteins, including their common variants, which can help expedite your antibody development.

Comprehensive cytokine targets including interleukins, growth factors, chemokines, TNFs, etc. are expressed by HEK293 to ensure their natural structure. Their high purity is verified by SDS-PAGE/HPLC/SEC-MALS and high bioactivity is verified by ELISA/SPR/BLI.

Aneuro provides innovative solutions for neuroscience research. Recombinant proteins, neural factors, pre-formed fibrils, electrophysiological electrodes, as well as Organoid Toolbox all in Aneuro aiming to advance neuroscience research, develop therapeutic interventions, and improve diagnostic methods for neurological diseases.
This web search service is supported by Google Inc.
